《Cell Stem Cell》| Top 10 Breakthrough Advances in Stem Cell Therapy in 2025
This article is a review of pluripotent stem cell-derived therapies in clinical trials, titled "Pluripotent Stem Cell-Derived Therapies in Clinical Trials: 2025 Update". It provides a detailed review of the progress of human pluripotent stem cells (IPSCs/CMCs) in clinical trials since their first isolation, with a focus on clinical safety and efficacy data, and offers a comprehensive summary of ongoing and completed clinical trials.
Key conclusions and findings:
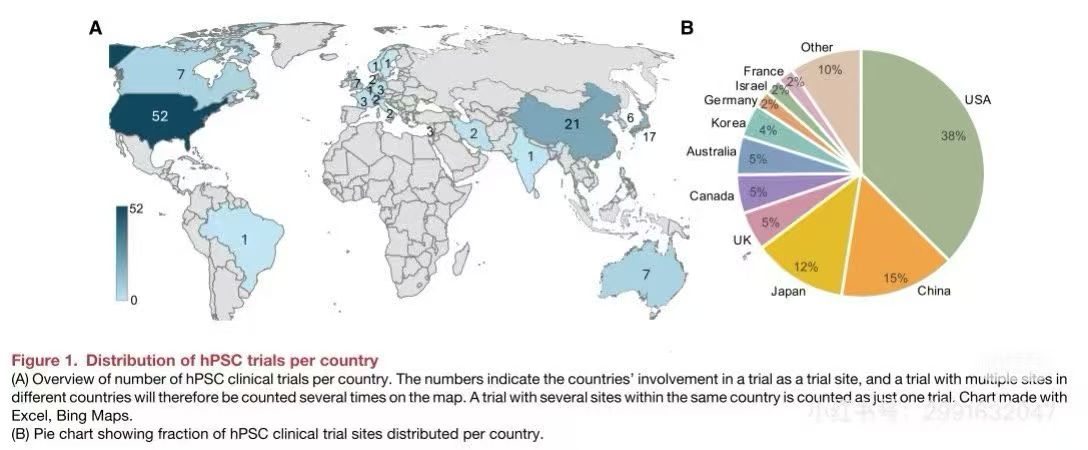
Overview of clinical trials: As of December 2024, there are 115 globally approved clinical trials involving 83 products, with over 1,200 patients treated and a cumulative use of more than 1,011 cells. No widespread safety issues have been identified so far.
Treatment of eye diseases: At least 21 products have entered clinical trials for eye diseases, including retinal pigment epithelial (RPE) replacement therapy, photoreceptors, corneal epithelial and endothelial cells, etc.
Central nervous system diseases: Parkinson's disease (PD) is a major research direction, involving dopaminergic neuron replacement therapy. In addition, there are clinical trials for spinal cord injury, epilepsy and other diseases.
Heart diseases: Multiple trials have tested the therapeutic effect of hPSC-derived cardiomyocytes on myocardial infarction and chronic heart failure.
Diabetes treatment: IPSCs/CMCs-derived islet cell products have shown good efficacy in clinical trials, with some patients achieving insulin independence.
Immune and blood diseases: CMCs-derived natural killer (NK) cells and T cell products have shown potential in the treatment of blood cancers.
Liver disease treatment: CMCs-derived pluripotent liver cells have shown excellent phagocytic ability in the initial treatment of liver cancer.